What is the Adaptive Designs for Clinical Trials of Drugs and Biologics Guidance for Industry. The 33-page guidance which finalizes a draft version released for comment in September 2018 and replaces an earlier guidance from 2010 sets out FDAs recommendations on adaptive trial design principles and the information FDA will review from adaptive studies submitted as part of investigational new drug applications INDs new drug applications. Adaptive design clinical trials for drugs and biologics
Adaptive Design Clinical Trials For Drugs And Biologics, 51 rm2201 Silver Spring MD 20993-0002 Tel 301-796-3400. Guidance for Industry Adaptive Design Clinical Trials for Drugs and Biologics Additional copies are available from. The concepts contained in this guidance are also useful for. FDA is a clinical trial design that allows for prospectively planned modifications to one or more aspects of the design based on accumulating data from subjects in the trial 1 Adaptive design characteristics include modifying an ongoing clinical trial in accordance with predetermined rules based on data from interim analyses.
 Clinical Trial Designs Basket Umbrella Platform Trial Designs Part Ii Credevo Articles From credevo.com
Clinical Trial Designs Basket Umbrella Platform Trial Designs Part Ii Credevo Articles From credevo.com
FDA has published a draft guidance on Adaptive Design Clinical Trials for Drugs and Biologics which gives regulatory guidance on methodological issues in exploratory and confirmatory clinical trials planned with an adaptive design. Today the Food and Drug Administration FDA issued final guidance for industry entitled Adaptive Designs for Clinical Trials of Drugs and Biologics Adaptive design clinical trials allow for prospectively planned modifications to one or more aspects of the design. The US Food and Drug Administration FDA last week finalized guidance on adaptive clinical trial designs for drugs and biologicsThis document provides guidance to sponsors and applicants submitting investigational new drug applications INDs new drug applications NDAs biologics licensing applications BLAs or supplemental applications on. Adaptive Design Clinical Trials for Drugs and Biologics Guidance for Industry December 2019 Download the Final Guidance Document Read the Federal Register Notice Final Level 1 Guidance.
FDA has published a draft guidance on Adaptive Design Clinical Trials for Drugs and Biologics which gives regulatory guidance on methodological issues in exploratory and confirmatory clinical trials planned with an adaptive design.
Read another article:
What is the Adaptive Designs for Clinical Trials of Drugs and Biologics Guidance for Industry. Adaptive designs for exploratory clinical trials deal mainly with. In 2010 the US Food and Drug Administration FDA released a draft guidance Guidance for Industry. Adaptive design as defined by the US. For the purposes of this guidance an adaptive design is defined as a clinical trial design that allows for prospectively planned modifications to one or more aspects of the design based on accumulating data from subjects in the trial.
 Source: clinicaltherapeutics.com
Source: clinicaltherapeutics.com
The clinical trial landscape has changed since 2010 regards the FDAs stance on. Adaptive designs are applicable to both exploratory and confirmatory clinical trials. This guidance finalizes the draft guidance entitled Adaptive Designs for Clinical Trials of Drugs and Biologics issued in October 2018. In 2010 the US Food and Drug Administration FDA released a draft guidance Guidance for Industry. The Evolution Of Master Protocol Clinical Trial Designs A Systematic Literature Review Clinical Therapeutics.
 Source: ris.world
Source: ris.world
The clinical trial landscape has changed since 2010 regards the FDAs stance on. The US Food and Drug Administration FDA last week finalized guidance on adaptive clinical trial designs for drugs and biologicsThis document provides guidance to sponsors and applicants submitting investigational new drug applications INDs new drug applications NDAs biologics licensing applications BLAs or supplemental applications on. FDA issues guidance for industry on adaptive designs for clinical trials of drugs and biologics. The concepts contained in this guidance are also useful for. Usa Adaptive Designs For Clinical Trials Of Drugs And Biologics Ris World.
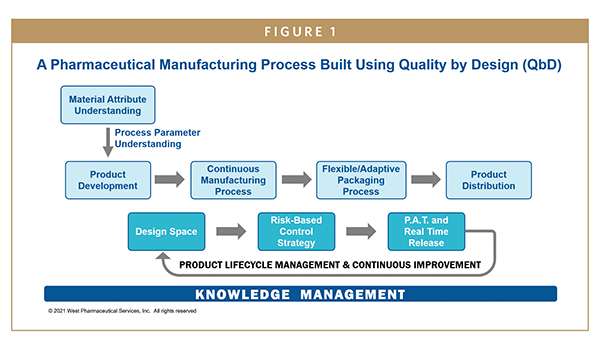 Source: drug-dev.com
Source: drug-dev.com
For the purposes of this guidance an adaptive design is defined as a clinical trial design that 44 allows for prospectively planned modifications to one or more aspects of the design based on. The 33-page guidance which finalizes a draft version released for comment in September 2018 and replaces an earlier guidance from 2010 sets out FDAs recommendations on adaptive trial design principles and the information FDA will review from adaptive studies submitted as part of investigational new drug applications INDs new drug applications. Today the Food and Drug Administration FDA issued final guidance for industry entitled Adaptive Designs for Clinical Trials of Drugs and Biologics Adaptive design clinical trials allow for prospectively planned modifications to one or more aspects of the design. Office of Communication Division of Drug Information Center for Drug Evaluation and Research Food and Drug Administration 10903 New Hampshire Ave Bldg. Drug Development Simplifying The Drug Development Journey.
 Source: resmedjournal.com
Source: resmedjournal.com
Adaptive designs for exploratory clinical trials deal mainly with. The primary focus of this guidance is on adaptive designs for clinical trials intended to support the effectiveness and safety of drugs. FDA has published a draft guidance on Adaptive Design Clinical Trials for Drugs and Biologics which gives regulatory guidance on methodological issues in exploratory and confirmatory clinical trials planned with an adaptive design. Specifically sponsors will be able to design trials that can be modified at preplanned stages with less risk of compromising the integrity of the data and any conclusions. In Line Treatments And Clinical Initiatives To Fight Against Covid 19 Outbreak Respiratory Medicine.
 Source: sciencedirect.com
Source: sciencedirect.com
The guidance provides information to. The primary focus of this guidance is on adaptive designs for clinical trials intended to support the effectiveness and safety of drugs. This guidance finalizes the draft guidance of the same title issued on October 1 2018 83 FR 49400. Specifically sponsors will be able to design trials that can be modified at preplanned stages with less risk of compromising the integrity of the data and any conclusions. Phase Ii Trials In Drug Development And Adaptive Trial Design Sciencedirect.
 Source: jdsjournal.com
Source: jdsjournal.com
The primary focus of this guidance is on adaptive designs for clinical trials intended to support the effectiveness and safety of drugs. FDA has published a draft guidance on Adaptive Design Clinical Trials for Drugs and Biologics which gives regulatory guidance on methodological issues in exploratory and confirmatory clinical trials planned with an adaptive design. The clinical trial landscape has changed since 2010 regards the FDAs stance on. Guidance for Industry Adaptive Design Clinical Trials for Drugs and Biologics Additional copies are available from. Short Term Effectiveness Of Biologics In Patients With Moderate To Severe Plaque Psoriasis A Systematic Review And Network Meta Analysis Journal Of Dermatological Science.
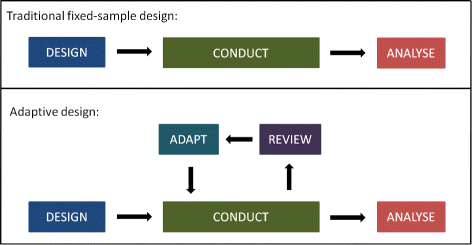 Source: bmcmedicine.biomedcentral.com
Source: bmcmedicine.biomedcentral.com
FDA issues guidance for industry on adaptive designs for clinical trials of drugs and biologics. Adaptive designs are applicable to both exploratory and confirmatory clinical trials. The US Food and Drug Administration FDA last week finalized guidance on adaptive clinical trial designs for drugs and biologicsThis document provides guidance to sponsors and applicants submitting investigational new drug applications INDs new drug applications NDAs biologics licensing applications BLAs or supplemental applications on. Office of Communication Division of Drug Information Center for Drug Evaluation and Research Food and Drug Administration 10903 New Hampshire Ave Bldg. Adaptive Designs In Clinical Trials Why Use Them And How To Run And Report Them Bmc Medicine Full Text.
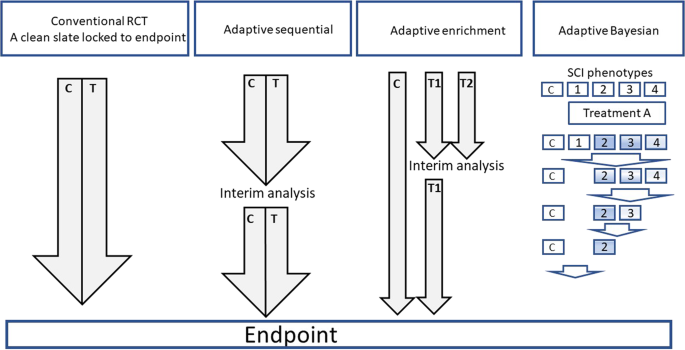 Source: nature.com
Source: nature.com
This guidance finalizes the draft guidance of the same title issued on October 1 2018 83 FR 49400. 51 rm2201 Silver Spring MD 20993-0002 Tel 301-796-3400. The US Food and Drug Administration FDA last week finalized guidance on adaptive clinical trial designs for drugs and biologicsThis document provides guidance to sponsors and applicants submitting investigational new drug applications INDs new drug applications NDAs biologics licensing applications BLAs or supplemental applications on. Guidance for Industry Adaptive Design Clinical Trials for Drugs and Biologics Additional copies are available from. Adaptive Trial Designs For Spinal Cord Injury Clinical Trials Directed To The Central Nervous System Spinal Cord.
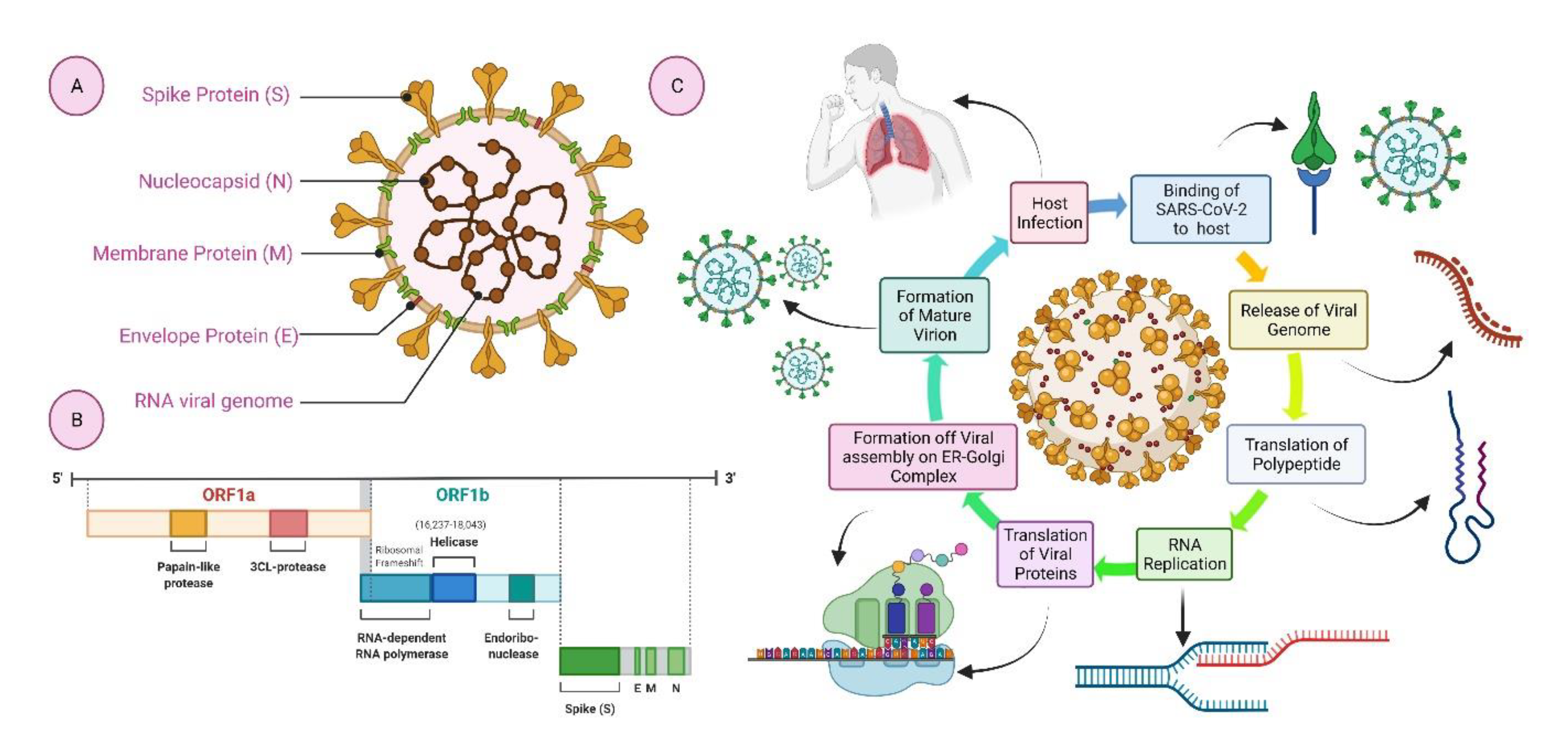 Source: mdpi.com
Source: mdpi.com
By casting dose finding as a Bayesian model selection problem we propose an adaptive design by simultaneously incorporating the toxicity and efficacy outcomes to select the optimal biological dose OBD in phase III clinical trials. Today the Food and Drug Administration FDA issued final guidance for industry entitled Adaptive Designs for Clinical Trials of Drugs and Biologics Adaptive design clinical trials allow for prospectively planned modifications to one or more aspects of the design. What is Adaptive Design Clinical Trial. The primary focus of this guidance is on adaptive designs for clinical trials intended to support the effectiveness and safety of drugs and biological products. Biologics Free Full Text Nucleic Acid Vaccines For Covid 19 A Paradigm Shift In The Vaccine Development Arena Html.
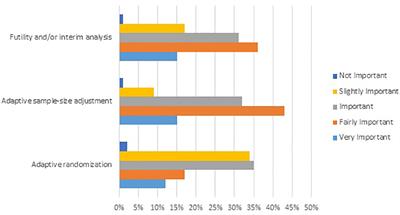 Source: frontiersin.org
Source: frontiersin.org
This guidance finalizes the draft guidance entitled Adaptive Designs for Clinical Trials of Drugs and Biologics issued in October 2018. The guidance describes important principles for designing conducting and reporting the results from an adaptive clinical trial. This comment summarizes the discussion within the joint working group Adaptive Designs and Multiple. The primary focus of this guidance is on adaptive designs for clinical trials intended to support the effectiveness and safety of drugs. Frontiers Value Of Adaptive Trials And Surrogate Endpoints For Clinical Decision Making In Rare Cancers Oncology.
 Source: valueinhealthjournal.com
Source: valueinhealthjournal.com
This guidance finalizes the draft guidance entitled Adaptive Designs for Clinical Trials of Drugs and Biologics issued in October 2018. The US Food and Drug Administration FDA last week finalized guidance on adaptive clinical trial designs for drugs and biologicsThis document provides guidance to sponsors and applicants submitting investigational new drug applications INDs new drug applications NDAs biologics licensing applications BLAs or supplemental applications on. Adaptive Design Clinical Trials for Drugs and Biologics Guidance for Industry December 2019 Download the Final Guidance Document Read the Federal Register Notice Final Level 1 Guidance. FDA has published a draft guidance on Adaptive Design Clinical Trials for Drugs and Biologics which gives regulatory guidance on methodological issues in exploratory and confirmatory clinical trials planned with an adaptive design. A Review Of Clinical Trials With An Adaptive Design And Health Economic Analysis Value In Health.
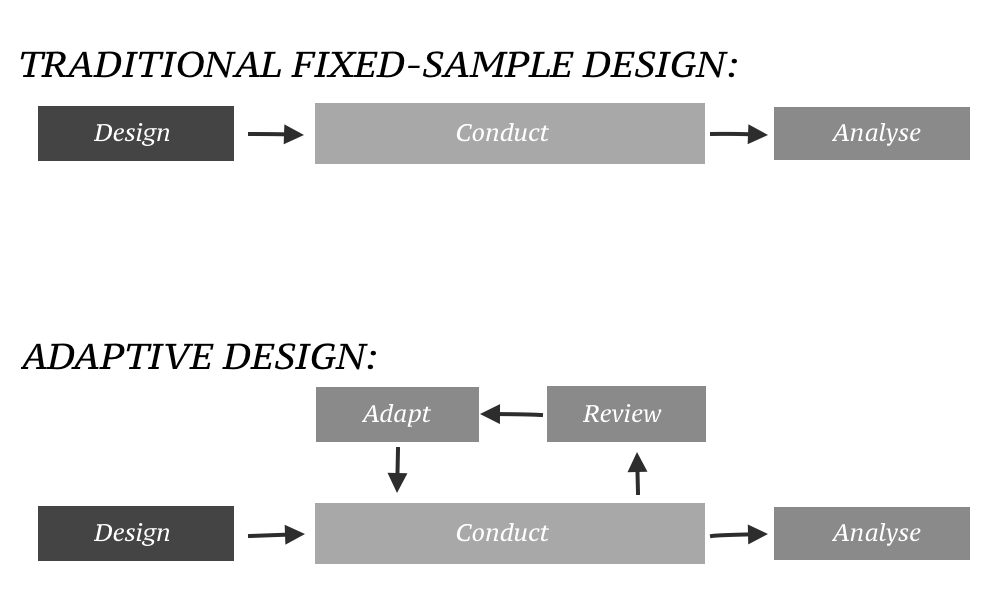 Source: en.wikipedia.org
Source: en.wikipedia.org
What is the Adaptive Designs for Clinical Trials of Drugs and Biologics Guidance for Industry. For the purposes of this guidance an adaptive design is defined as a clinical trial design that allows for prospectively planned modifications to one or more aspects of the design based on accumulating data from subjects in the trial. The primary focus of this guidance is on adaptive designs for clinical trials intended to support the effectiveness and safety of drugs and biological products. FDA is a clinical trial design that allows for prospectively planned modifications to one or more aspects of the design based on accumulating data from subjects in the trial 1 Adaptive design characteristics include modifying an ongoing clinical trial in accordance with predetermined rules based on data from interim analyses. Adaptive Design Medicine Wikipedia.
 Source: cell.com
Source: cell.com
This guidance finalizes the draft guidance entitled Adaptive Designs for Clinical Trials of Drugs and Biologics issued in October 2018. This guidance finalizes the draft guidance of the same title issued on October 1 2018 83 FR 49400. For the purposes of this guidance an adaptive design is defined as a clinical trial design that allows for prospectively planned modifications to one or more aspects of the design based on accumulating data from subjects in the trial. FDA has published a draft guidance on Adaptive Design Clinical Trials for Drugs and Biologics which gives regulatory guidance on methodological issues in exploratory and confirmatory clinical trials planned with an adaptive design. Clinical Development Of Gene Therapies The First Three Decades And Counting Molecular Therapy Methods Clinical Development.
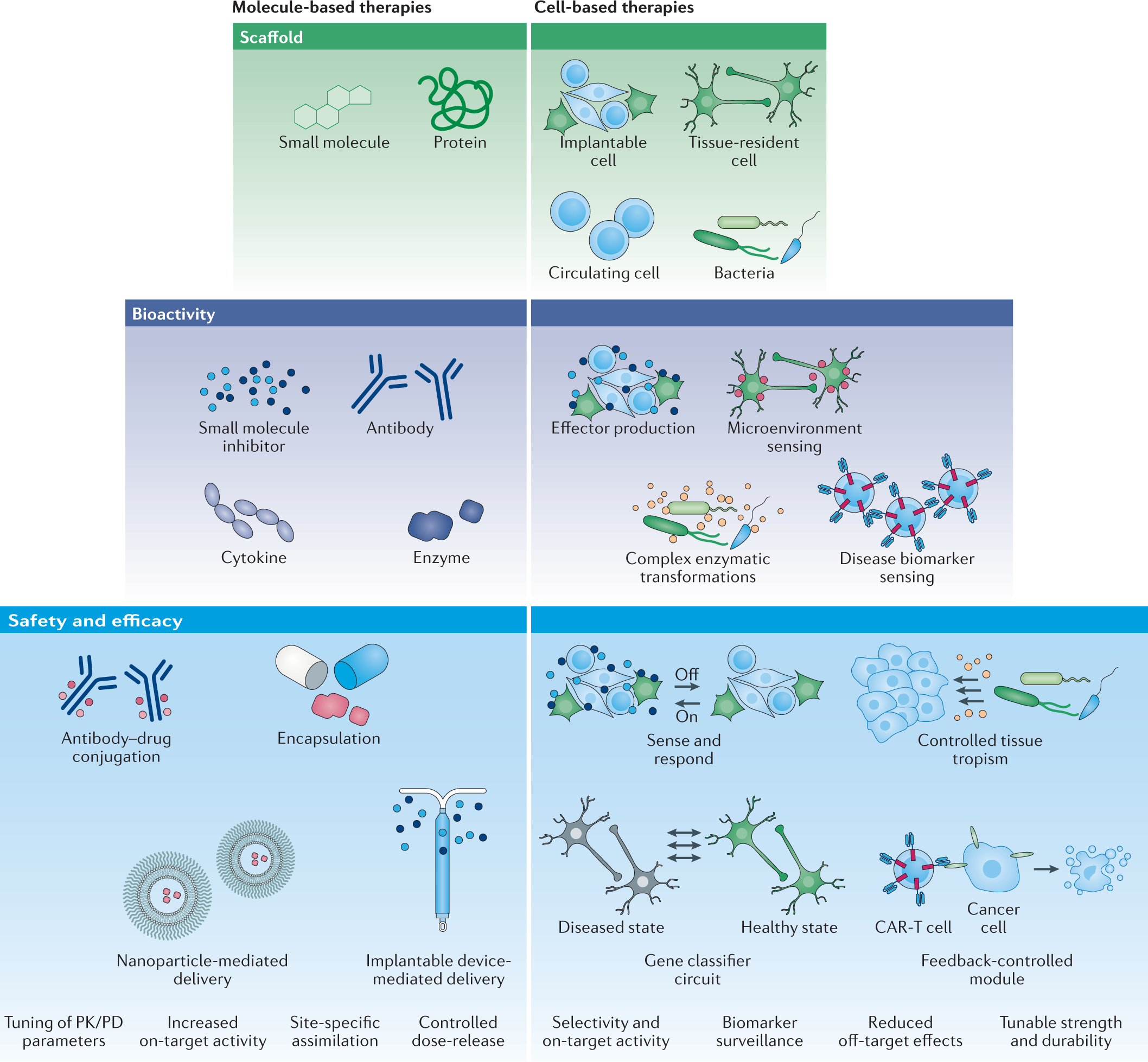 Source: nature.com
Source: nature.com
What is Adaptive Design Clinical Trial. Office of Communication Division of Drug Information Center for Drug Evaluation and Research Food and Drug Administration 10903 New Hampshire Ave Bldg. The purpose is to. Specifically sponsors will be able to design trials that can be modified at preplanned stages with less risk of compromising the integrity of the data and any conclusions. Engineering Living Therapeutics With Synthetic Biology Nature Reviews Drug Discovery.
 Source: acsjournals.onlinelibrary.wiley.com
Source: acsjournals.onlinelibrary.wiley.com
Adaptive designs for exploratory clinical trials deal mainly with. This guidance will replace the 2010 draft guidance for industry Adaptive Design Clinical Trials for Drugs and Biologics. What is the Adaptive Designs for Clinical Trials of Drugs and Biologics Guidance for Industry. Today the Food and Drug Administration FDA issued final guidance for industry entitled Adaptive Designs for Clinical Trials of Drugs and Biologics Adaptive design clinical trials allow for prospectively planned modifications to one or more aspects of the design. Clinical Trial Design Past Present And Future In The Context Of Big Data And Precision Medicine Li 2020 Cancer Wiley Online Library.







